Atomic Number of Tungsten is 74.
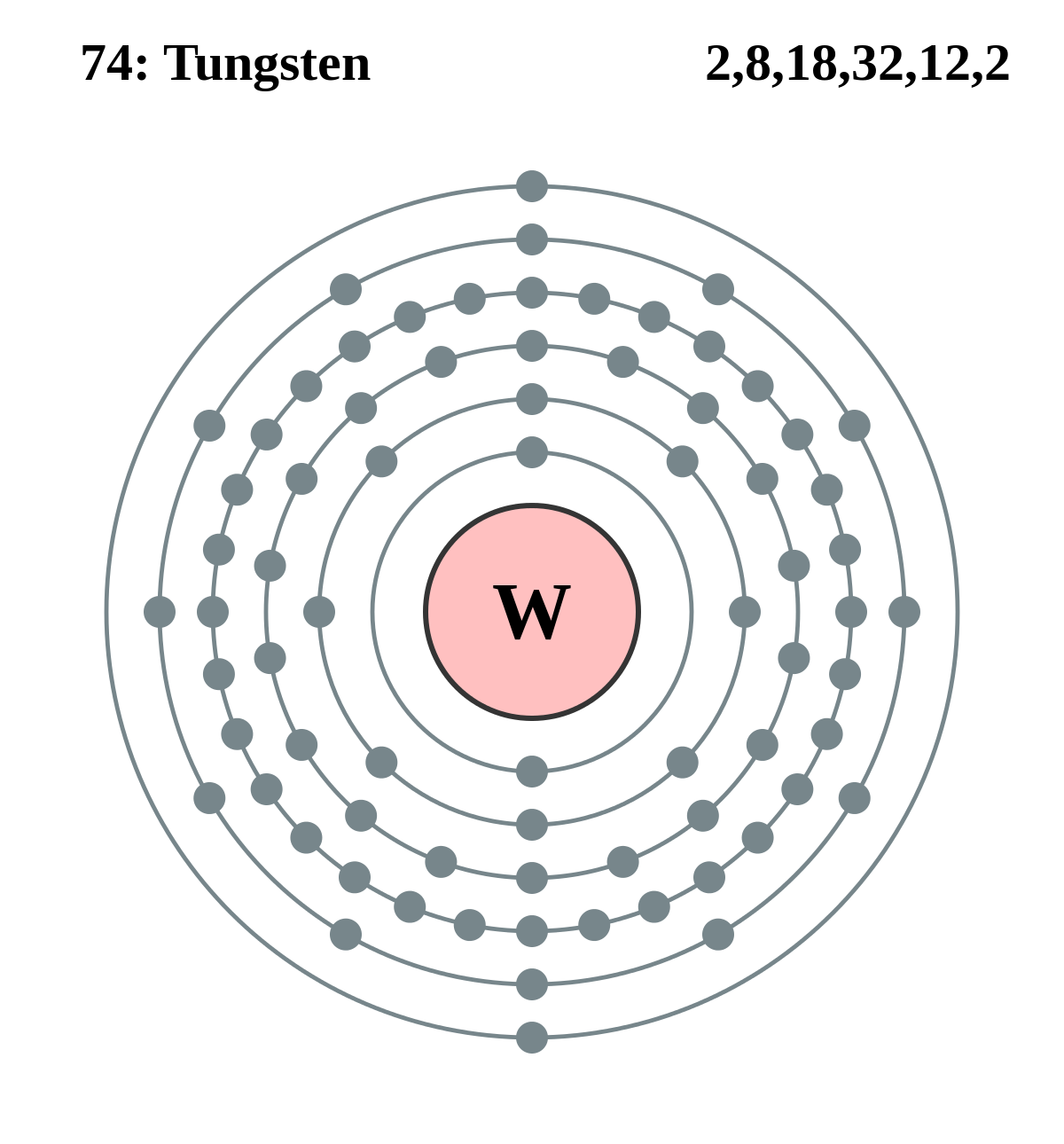
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isolated as a metal in 1783. Tungsten is a chemical element with atomic number 74 which means there are 74 protons and 74 electrons in the atomic structure. The chemical symbol for Tungsten is W. Atomic Mass of Tungsten Atomic mass of Tungsten is 183.84 u. W I Ground State 1s 2 2s 2 2p 6 3s 2 3p 6 3d 1 0 4s 2 4p 6 4d 1 0 4f 1 4 5s 2 5p 6 5d 4 6s 2 5 D 0 Ionization energy 63427.7 cm-1 (7.86403 eV) Ref. CS96 W II Ground State 1s 2 2s 2 2p 6 3s 2 3p 6 3d 1 0 4s 2 4p 6 4d 1 0 4f 1 4 5s 2 5p 6 5d 4 6s 6 D 1 / 2 Ionization energy 130000 cm-1 (16.1 eV) Ref.
Chemical symbol for Tungsten is W. Number of protons in Tungsten is 74. Atomic weight of Tungsten is 183.84 u or g/mol. Melting point of Tungsten is 3407 °C and its the boiling point is 5927 °C.
» Boiling Point» Melting Point» Abundant» State at STP» Discovery Year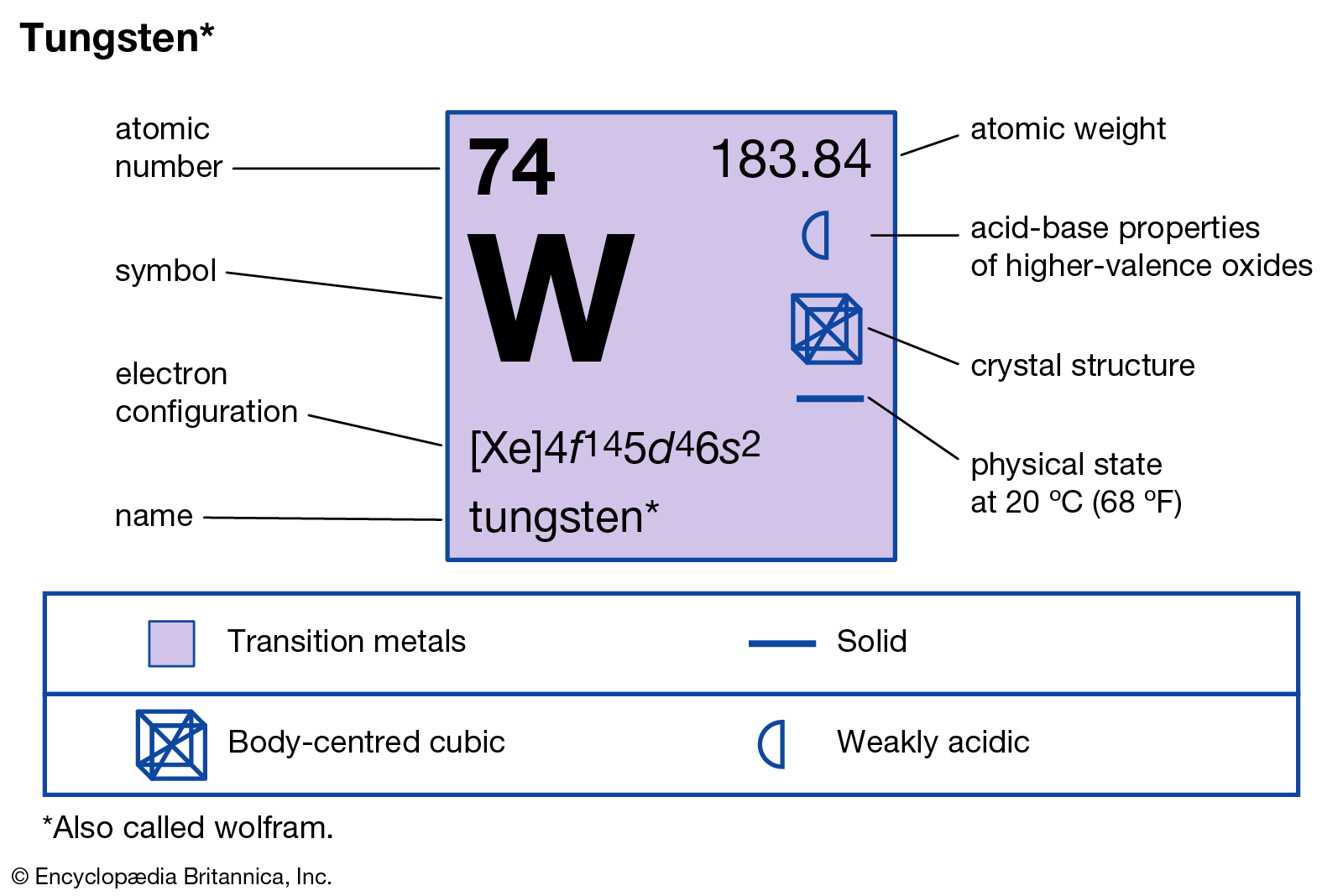
About Tungsten
This chemical element was discovered at the end of the 18th century and named after a Swedish expression meaning heavy stone. It is a strong shiny light grey color metal, and it is one of the heaviest metals which have a certain importance for life since some types of bacteria have tungsten in their enzymes. It is possible to obtain this metal from minerals, and one more way to obtain it is from its compounds with carbon or oxygen. Tungsten is used in light bulbs production, and it can form very strong alloys with other metals, for further use in producing elements for welding, heating, etc. Chemical compounds of this element have applications in oil and petroleum refining, X-ray tubes, drilling and cutting tools, etc.
Properties of Tungsten Element
| Atomic Number (Z) | 74 |
|---|---|
| Atomic Symbol | W |
| Group | 6 |
| Period | 6 |
| Atomic Weight | 183.84 u |
| Density | 19.25 g/cm3 |
| Melting Point (K) | 3695 K |
| Melting Point (℃) | 3407 °C |
| Boiling Point (K) | 5828 K |
| Boiling Point (℃) | 5927 °C |
| Heat Capacity | 0.132 J/g · K |
| Abundance | 1.3 mg/kg |
| State at STP | Solid |
| Occurrence | Primordial |
| Description | Transition metal |
| Electronegativity (Pauling) χ | 2.36 |
| Ionization Energy (eV) | 7.864 |
| Atomic Radius | 135pm |
| Covalent Radius | 146pm |
| Valence Electrons | 2 |
| Year of Discovery | 1783 |
| Discoverer | Gebrüder de Elhuyar |
What is the Boiling Point of Tungsten?
Tungsten Atomic Mass In Grams
Tungsten boiling point is 5927 °C. Boiling point of Tungsten in Kelvin is 5828 K.
What is the Melting Point of Tungsten?
Tungsten melting point is 3407 °C. Melting point of Tungsten in Kelvin is 3695 K.
How Abundant is Tungsten?
Abundant value of Tungsten is 1.3 mg/kg.
What is the State of Tungsten at Standard Temperature and Pressure (STP)?
State of Tungsten is Solid at standard temperature and pressure at 0℃ and one atmosphere pressure.
When was Tungsten Discovered?
Tungsten Atomic Mass Grams Per Mole
Tungsten was discovered in 1783.
